Home | About | Faculty | Calendar | Facilities | Graduate program | Contact | Apply
This page is optimized for viewing with javascript.
Mike Harms

Assistant Professor, Chemistry and Biochemistry
Member, IMB
Ph.D. Johns Hopkins University
B.S. Oregon State University
Email
Lab website
Office: Willamette Hall Room 340A
Office Phone: 541-346-9002
Lab: Willamette Hall Room 340
Lab Phone: 541-346-9003
Loading profile for David Garcia
Research Interests
Cellular memories of a stressful past: encoding by epigenetic regulation of RNA and other mechanisms
A cell or organism's ability to adapt to new environments or stress has major impacts on its evolutionary success, and its potential to transform into diseased states. Genetically encoded traits store selected information that protects against numerous destabilizing forces, experienced over evolutionary timescales. Epigenetic traits can promote rapid phenotypic change, controlling how the complexly evolved genome is expressed upon stress or novel environmental signals.
Our lab aims to uncover, in molecular detail, epigenetic traits that influence how cells respond to stress and adapt to new environments. We mix molecularly focused analyses and high-throughput experiments in yeast to define the drivers of these stress/environment-responsive epigenetic states. We are interested in taking what we learn in yeast and exploring its conservation in metazoans, to understand how these traits can affect human health or influence disease.
RNA Modifying Enzymes with Prion-Like Properties
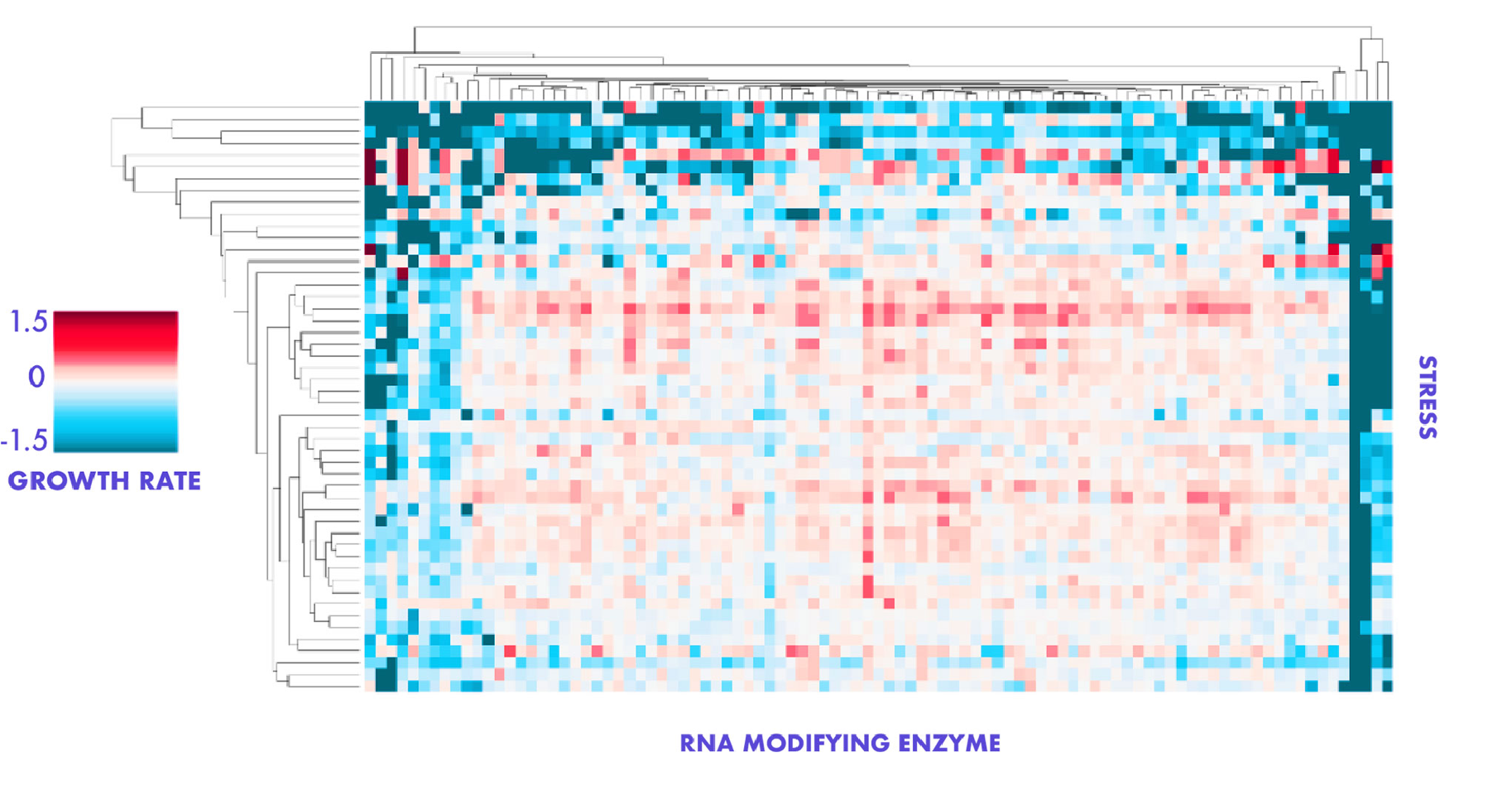
We place particular focus on regulators of RNA chemical modifications or structure. RNA modifying enzymes (RMEs) can influence expression of numerous genes simultaneously, through molecularly definable rules. Through a large phenotypic screen, based on transient perturbation of more than seventy RMEs and two-dozen other RBPs, we discovered numerous examples of RMEs driving prion-like, heritable growth states. These states influenced fundamental traits such as proliferation, cell size, lifespan, and growth in a variety of stresses. Our goals are to understand the causes and effects of this type of regulation, by connecting long-lasting, heritable changes in RNA regulation to their consequent effects on cell growth across varying environments.
Stress-Induced Epigenetic States
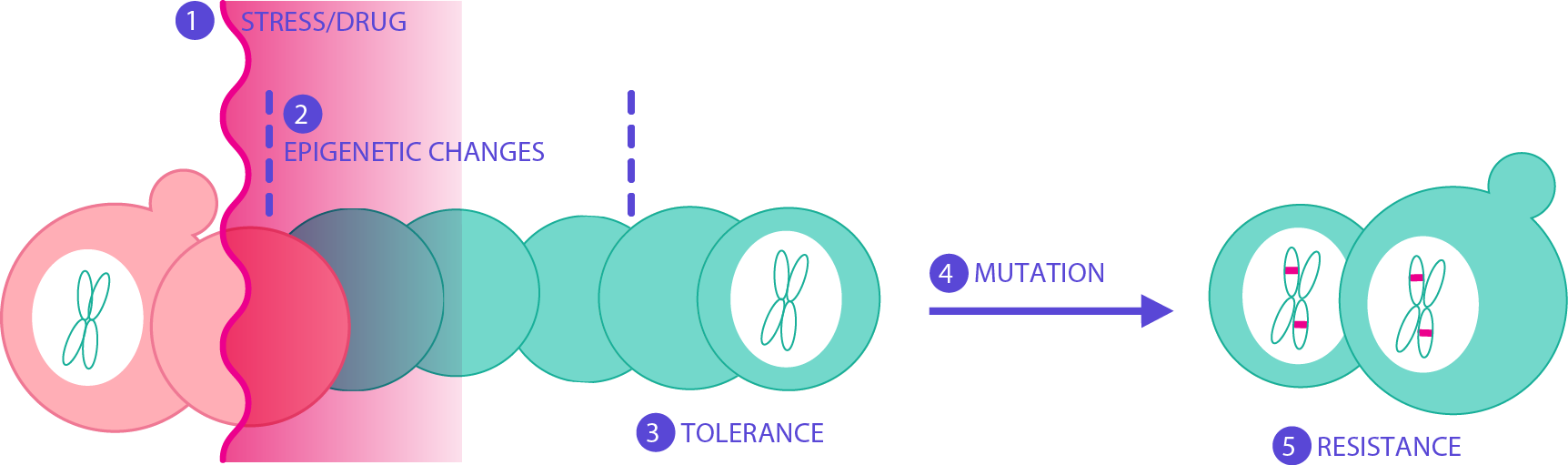
We are also interested in the ability of molecules—both natural and man-made—and environmental stresses to induce new heritable growth states. We previously showed the possibility of identifying environmental molecules responsible for inducing adaptive prions in yeast (Garcia and Dietrich et al., 2016). We then approached this question from a different angle, executing a large phenotypic screen, testing many known molecules and discovering their potential to induce new heritable growth states. We want to identify the genetic and molecular basis of these long-lived states.
Please check out the Garcia lab website for more details.
Recent publications
(pulled from pubmed)
Recent publications
(pulled from pubmed)
Harms MJ, Thornton JW
Nature 2014 Aug 14;512(7513):203-7
Hart KM, Harms MJ, Schmidt BH, Elya C, Thornton JW, Marqusee S
PLoS Biol 2014 Nov;12(11):e1001994
Harms MJ, Eick GN, Goswami D, Colucci JK, Griffin PR, Ortlund EA, Thornton JW
Proc Natl Acad Sci U S A 2013 Jul 9;110(28):11475-80
Harms MJ, Thornton JW
Nat Rev Genet 2013 Aug;14(8):559-71
Eick GN, Colucci JK, Harms MJ, Ortlund EA, Thornton JW
PLoS Genet 2012;8(11):e1003072
Harms MJ, Schlessman JL, Sue GR, García-Moreno B
Proc Natl Acad Sci U S A 2011 Nov 22;108(47):18954-9
Harms MJ, Thornton JW
Curr Opin Struct Biol 2010 Jun;20(3):360-6
Bridgham JT, Eick GN, Larroux C, Deshpande K, Harms MJ, Gauthier ME, Ortlund EA, Degnan BM, Thornton JW
PLoS Biol 2010 Oct 5;8(10)
Harms MJ, Castañeda CA, Schlessman JL, Sue GR, Isom DG, Cannon BR, García-Moreno E B
J Mol Biol 2009 May 29;389(1):34-47
Harms MJ, Schlessman JL, Chimenti MS, Sue GR, Damjanović A, García-Moreno B
Protein Sci 2008 May;17(5):833-45
Harms MJ, Wilmarth PA, Kapfer DM, Steel EA, David LL, Bächinger HP, Lampi KJ
Protein Sci 2004 Mar;13(3):678-86